 |
 |
- Search
| Clin Shoulder Elb > Volume 26(4); 2023 > Article |
|
Abstract
Background
Methods
Results
NOTES
Author contributions
Conceptualization: JTH. Data curation: TYK. Formal analysis: YTK, TYK. Supervision: JTH. Validation: YTK. Writing–original draft: YTK, TYK. Writing–review & editing: JBL.
Fig. 1.

Fig. 2.
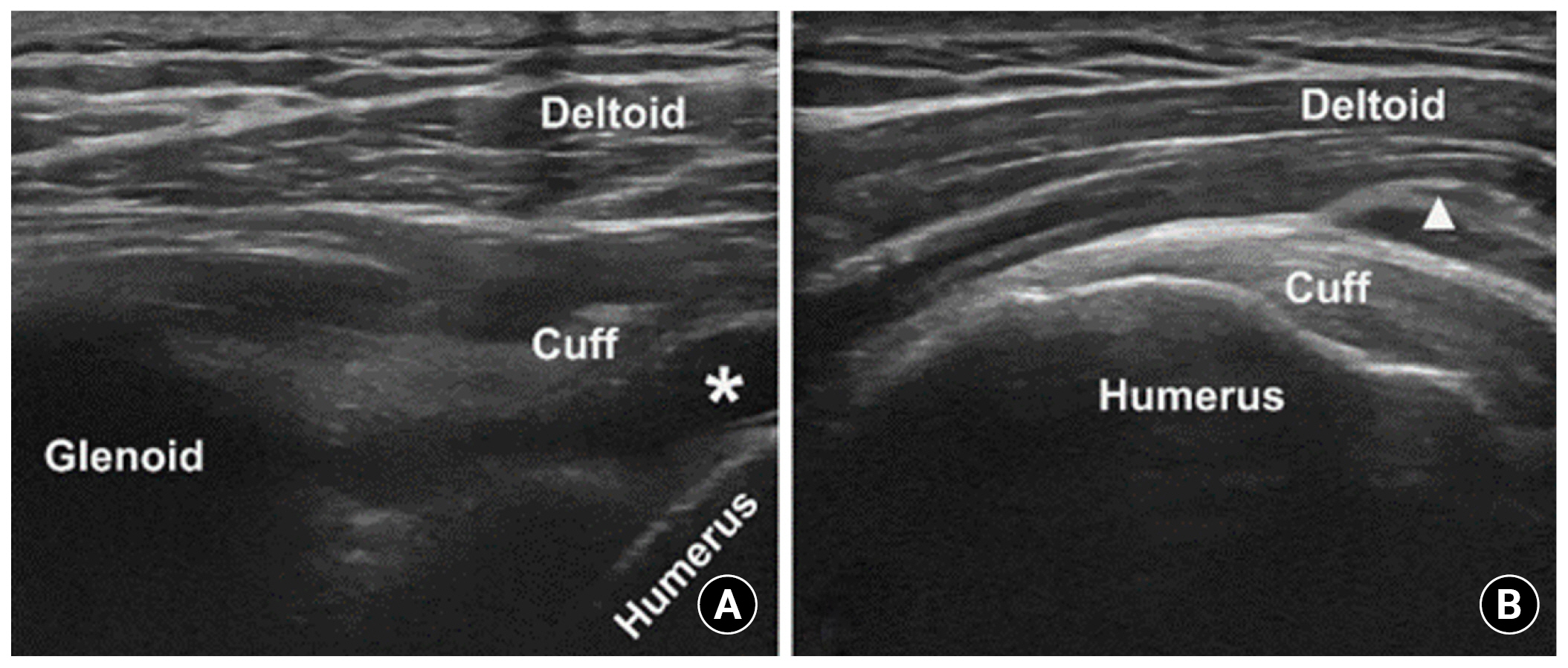
Fig. 3.

Table 1.
Values are presented as mean±standard deviation or number (%). GH group: ultrasound-guided glenohumeral steroid injection, SA group: ultrasound-guided subacromial steroid injection.
FE: forward elevation, ROM: range of motion, ER: external rotation at side, IR: internal rotation at 90° of abduction, pVAS: pain visual analog scale, ASES: American Shoulder and Elbow Surgeons, Constant: Constant-Murley.
Table 2.
| Variable | Pre-injection (n=24) | Final follow-up (n=24) | P-value* |
|---|---|---|---|
| GH group (n=24) | |||
| FE ROM (°) | 127.5±7.4 | 148.8±3.4 | <0.001 |
| ER at side ROM (°) | 53.8±13.1 | 76.9±13.0 | <0.001 |
| IR at 90° ROM (°) | 45.4±7.2 | 75.0±11.5 | <0.001 |
| pVAS score | 6.3±2.0 | 2.6±2.1 | <0.001 |
| ASES score | 46.1±18.4 | 74.5±19.0 | <0.001 |
| Constant score | 55.9±7.9 | 67.2±8.8 | <0.001 |
| SA group (n=24) | |||
| FE ROM (°) | 129.2±8.3 | 148.3±3.8 | <0.001 |
| ER at side ROM (°) | 54.6±10.8 | 76.5±8.0 | <0.001 |
| IR at 90° ROM (°) | 48.8±9.0 | 74.4±8.9 | <0.001 |
| pVAS score | 6.0±1.2 | 2.6±1.5 | <0.001 |
| ASES score | 53.0±8.9 | 77.1±12.9 | <0.001 |
| Constant score | 59.2±7.6 | 68.7±9.9 | <0.001 |
Values are presented as mean±standard deviation. GH group: ultrasound-guided glenohumeral steroid injection, SA group: ultrasound-guided subacromial steroid injection.
FE: forward elevation, ROM: range of motion, ER: external rotation at side, IR: internal rotation at 90˚ of abduction, pVAS: pain visual analog scale, ASES: American Shoulder and Elbow Surgeons, Constant: Constant-Murley.
Table 3.
| GH vs. SA group |
P-value |
|||||
|---|---|---|---|---|---|---|
| ΔFE ROM | ΔER ROM | ΔIR ROM | ΔpVAS | ΔASES | ΔConstant | |
| 3 wk–Pre | <0.001* | 0.012* | 0.002* | 0.637 | 0.768 | 0.078 |
| 7 wk–Pre | 0.082 | 0.112 | <0.001* | 0.730 | 0.558 | 0.046* |
| 13 wk–Pre | 0.288 | 0.402 | 0.129 | 0.333 | 0.366 | 0.228 |
GH group: ultrasound-guided glenohumeral steroid injection, SA group: ultrasound-guided subacromial steroid injection. Δ: amount of improvement=follow-up measurement subtracted by the pre-injection (Pre) measurement.
FE: forward elevation, ROM: range of motion, ER: external rotation at side, IR: internal rotation at 90° of abduction, pVAS: pain visual analog scale, ASES: American Shoulder and Elbow Surgeons, Constant: Constant-Murley.








