A review of chronic pectoralis major tears: what options are available?
Article information
Abstract
Rupture of the pectoralis major muscle typically occurs in the young, active male. Acute management of these injuries is recommended; however, what if the patient presents with a chronic tear of the pectoralis major? Physical exams and magnetic resonance imaging can help identify the injury and guide the physician with a plan for management. Nonoperative management is feasible, but is recommended for elderly, low-demand patients whose functional goals are minimal. Repair of chronic tears should be reserved for younger, healthier patients with high functional demands. Although operative management provides better functional outcomes, operative treatment of chronic pectoralis tears can be challenging. Tendon retraction, poor tendinous substance and quality of tissue, muscle atrophy, scar formation, and altered anatomy make direct repairs complicated, often necessitating auto- or allograft use. We review the various graft options and fixation methods that can be used when treating patients with chronic pectoralis major tears.
INTRODUCTION
The pectoralis major is the largest muscle of the anterior chest wall and functions to aid in internal rotation and adduction of the shoulder [1,2]. The origin of the muscle includes both a clavicular head with an attachment on the anterior surface of the medial clavicle and a sternocostal head with a broad attachment site that includes the anterior sternum, six superior costal cartilages, sternal end of the 6th rib, and the aponeurosis of the external oblique muscle. At its insertion, muscle fibers converge into a broad, flat bilaminate tendon that attaches at the lateral lip of the intertubercular sulcus of the humerus (Fig. 1). Tears of the pectoralis major muscle are relatively rare, occurring predominantly in young, active males (20–40 years old). The most common site of injury is at the tendon insertion (65%), followed by the musculotendinous junction (27%) [3-5].
Acute pectoralis major tears often present with a “pop” or “tearing” sensation associated with sudden pain in the medial aspect of the upper arm. Patients will have weakness with resisted adduction and internal rotation of the affected shoulder [6,7]. On examination, medial tendon retraction may result in a palpable defect of the anterior axillary fold which manifests as a prevalent diagnostic feature. Without acute treatment of the injury, the pectoralis major muscle and tendon may undergo significant muscle atrophy and retraction leading to an exaggerated absence of the anterior axillary fold [6]. Additionally, traction deformity characterized by bunching at the superolateral aspect of the sternal head origin is common [8,9].
Previous literature has suggested that early treatment of these injuries leads to improved clinical outcomes [10-13]. Retraction, poor tendinous substance, and tear location contribute to the challenging nature of chronic injuries and are likely related to inferior outcomes [8]. While recent reviews have evaluated surgical and conservative management for acute pectoralis major tears, recommendations for the management of chronic injuries remain scarce [1]. In this review we summarize the outcomes of various methods of management used in treatment of chronic pectoralis major tears.
CLASSIFICATION
Pectoralis major tendon tears were previously classified by Bak et al. [14] and Tietjen et al. [15] based on severity and location of injury (Table 1). However, these injuries are often categorized by chronicity, which has a considerable effect on management. While the definition of acute versus chronic pectoralis major tears remains ambiguous in the literature, in this review we define chronic tears as those presenting at least 6 weeks after initial injury (Table 2) [3,12,14-16].
IMAGING
Acute pectoralis major tears can typically be clinically diagnosed, but this may not be the case for chronic injuries. While bony abnormalities associated with these injuries are rare, radiographs of the shoulder and humerus may be beneficial to rule out bony avulsion or an associated fracture or dislocation in cases resulting from atypical mechanisms [8]. The addition of advanced imaging may be used to confirm diagnosis and aid in preoperative planning of chronic tears to determine if graft reconstruction is potentially beneficial [1,17].
Ultrasound may be used as a cost and time efficient method for diagnosis and can avoid delays in treatment when clinical signs are apparent and further work-up is time consuming or detrimental to the patient, as in cases where insurance authorization may be prolonged [18]. Using comparisons of injured and uninjured sides, normal hypoechoic muscle tissue can be identified and contrasted with hyperechoic hematoma formation, which may be present following injury. Retraction of the pectoralis major tendon may create a gap between the deltopectoral groove and coracobrachialis muscle that can also be identified [1].
Magnetic resonance imaging (MRI) is the imaging modality of choice for evaluating pectoralis major tears. The ability to distinguish between partial and complete tears regardless of chronicity is valuable for determining severity and guiding treatment recommendations [19,20]. In the acute setting, T1-weighted axial imaging has been found to be most effective in determining tears, while T2-weighted imaging has been found to be most effective in evaluation of chronic injuries [21]. Chronic tears may reveal low signal intensity indicating scarring and fibrosis [22]. MRI is often necessary to determine the need for graft reconstruction in the treatment of chronic injuries through evaluation of tissue quality and tendon retraction [1]. It is important to note that a traditional MRI sequence of the shoulder is not ideal for assessing pectoralis injuries, as the entire hemi-chest is required for complete evaluation [17,23]. Therefore, MRI imaging of the shoulder must be ordered with specific protocols if pectoralis injury is suspected, or the provider may opt to order an MRI of the chest to prevent confusion with protocol.
TREATMENT OPTIONS
Chronic pectoralis major tears can be treated nonoperatively or surgically, via repair or reconstruction depending on individualized patient and tear factors. Patient factors consist of age, activity level, medical comorbidities, history of anabolic steroids and certain antibiotics, and expectations. Tear morphology, such as chronicity, location, severity, and number of heads involved also plays a critical role in determining the optimal management for individual patients [1,3,13,24,25]. For chronic tears, it is essential to take a thorough history to understand why a patient is seeking medical care now. It is important to differentiate whether the patient did not seek medical care, if the injury was missed, or if it was previously diagnosed and treated with conservative management but the patient is unhappy with the outcome. Is the patient presenting due to cosmetic concerns, functional deficits, or pain? The answers to these questions may provide important insights on the patient’s acute condition and play influential roles in management.
NONOPERATIVE MANAGEMENT
Conservative management of chronic pectoralis major tears may be considered in patients of older age, less active lifestyles, poor compliance with postoperative rehabilitation, less extensive tears, and other medical comorbidities [1]. Nonoperative modalities may include physical therapy, lifestyle changes, and pain management. In our practice, we have utilized ultrasound, electrotherapy, and passive range of motion exercises early in the rehabilitation process with progression to stretching and light resistance exercises, eventually allowing resistance program training and athletic activity. Previous meta-analysis has demonstrated the superiority of surgical repair over conservation management, including improved functional score, proportion of shoulders gaining full isometric strength, change in isokinetic strength when compared to the contralateral shoulder, presence of resting deformity and overall satisfaction with cosmesis in patients who underwent surgery [6,14]. However, for low-demand patients, returning to their activities of daily living without pain is adequate, and therefore surgery may not be necessary. Surgery remains an option for cases where conservative management has not allowed the patient to achieve their functional goals.
OPERATIVE MANAGEMENT
Chronic pectoralis major tears present a unique challenge to orthopedic surgeons. Tendon retraction, poor tendinous substance and quality, tear location, muscle atrophy, scar formation, and altered anatomy lead to increased tension of the musculotendinous unit and make direct repairs more complicated in chronic injuries compared to acute injuries [1,8,13,16,23,26]. For these reasons, many surgeons will opt to treat chronic injuries nonoperatively. Although nonoperative management may be a viable option for certain patients, functional outcome scores are reported to be higher among patients who elect for surgical treatments [6,14].
When the musculotendinous unit of the pectoralis is unable to be adequately mobilized, autograft or allograft reconstruction may be required. The uses of many different graft types have been described in the literature, including bone-patellar tendon (BPT) autograft, gracilis-semitendinosus autograft, iliotibial band (ITB) autograft, semitendinosus allograft, fascia lata allograft, Achilles tendon allograft, and dermal allograft (Table 3) [8,13,16,26-33].
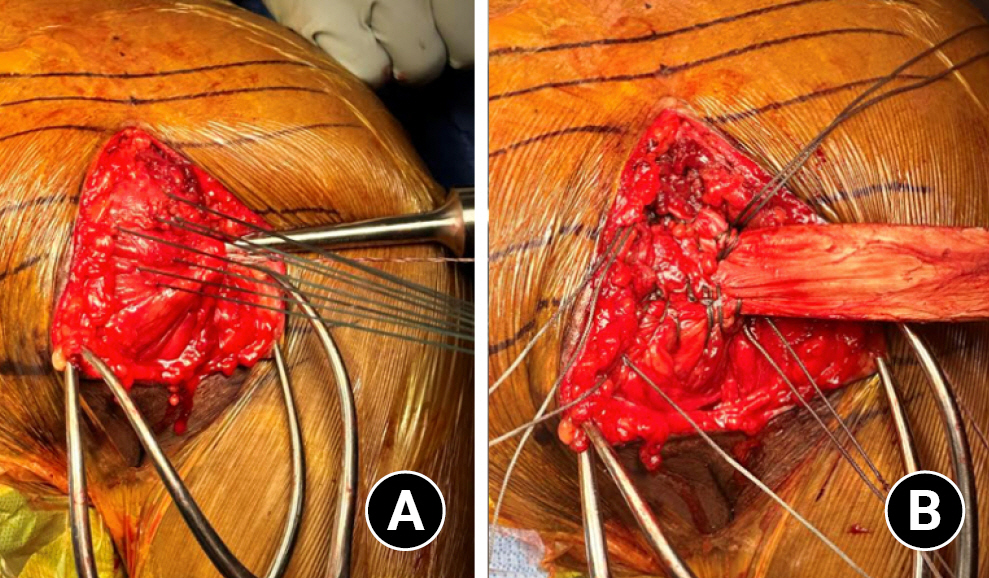
The decision to perform a reconstruction with a graft is typically made clinically, based upon the degree of pectoralis major retraction, but this often cannot be determined until all adhesions are released at the time of surgery (Fig. 2). Even MRI has not been found to be of significant use in deciding whether or not a graft for reconstruction is necessary [1,28]. As a result, it is recommended to have a back-up plan if standard end-to-end repair is not possible. There is a paucity of literature regarding the operative management of chronic pectoralis major tears. However, many graft types have been documented in the use of pectoralis major reconstruction, and as such, surgeons should be prepared to use one if necessary. Below we review the various techniques and graft options in the operative management of these injuries. Of note, the majority of the current literature describes use of a deltopectoral approach with suture anchors as a means of fixation to the humerus.
FIXATION METHODS
Suture anchor fixation has been used heavily in previous studies when performing reconstructions of chronic pectoralis major tears. Other documented fixation methods include cortical screw fixation, humeral drill holes, and cortical buttons. There is little research comparing these methods. Previous research has shown the use of various graft options, as well as several suture techniques. Allograft appears to be the more popular option, but there is a lack of research comparing allograft to autograft fixation on patient outcomes. This also applies to suture techniques incorporating both the anchor to the graft and the graft to the native pectoralis stump. Previous authors have documented using the Krackow technique, the Pulvertaft technique [34], the modified whip-stitch, and the modified Becker technique [35], with the Krackow being used most commonly.
GRAFT CHOICE
ITB Autograft
Decker et al. [33] described a case of a chronic pectoralis major tendon tear reconstructed using an ITB autograft. The free ITB graft was folded over the lateral tendon-deficient end of the pectoralis major muscle with 5–6 cm of graft covering both the anterior and posterior aspects of the distal muscle belly. The remaining 2 cm of graft was utilized to reconstruct the all-tendinous portion of the pectoralis major. Secure fixation was achieved by sequential passing of sutures anterior to posterior through the graft and muscle. Three double-loaded anchors were deployed equally spaced along the humeral insertion site. Both limbs of one of the sutures from each of the three suture anchors were passed in simple mattress fashion through the respective site on the folded end of the graft. A locking modified-Krackow technique was used to pass a single limb from each remaining suture anchor in a lateral to medial fashion along the free folded edge of the tendon graft, while the opposite limb was passed posteriorly to anteriorly at the lateral edge. At 2-year follow up, the patient had recovered full functional range of motion (ROM), strength and appearance, along with “excellent” outcomes as per the clinical outcome scale classified by Bak et al. [14,33].
Hamstring Allograft
Long et al. [30] described a case series of 6 patients who sustained a pectoralis major tendon tear that was repaired using a semitendinosus allograft with number 2 polyethylene suture that was placed in whip-stitch fashion in each end of the graft. By use of the Pulvertaft technique [34], the suture limbs from the graft were weaved through the pectoralis major muscle belly leaving three distinct limbs on the most lateral aspect of the muscle. This attachment was reinforced with a Braided number 2 polyethylene suture that was placed through the middle limb, and multiple additional sutures were used to tie the tendon limbs together. The three distinct limbs that were left untied at the most lateral aspect of the pectoralis muscle were passed through 3 G2 Mitek suture anchors (DePuy Synthes) placed at the anatomical footprint on the humerus. At 1-year follow-up, patients had excellent subjective and functional outcomes, as measured by American Shoulder and Elbow Surgeons score (average, 98.3; range, 95–100), Constant score (98 for all patients), and visual analog scale (VAS) (average, 0.67; range, 0–1).
Schachter et al. [31] used a standard quadruple-strand hamstring graft to bridge a 2-cm residual gap after a chronic pectoralis tendon tear was maximally mobilized. The gracilis and semitendinosus tendons were harvested and the tendons were looped to form a standard quadruple-strand graft. The looped end of the graft was stitched together utilizing number 2 FiberWire (Arthrex) which were then passed through humeral drill holes and tied over the remaining bone bridge, while the four-strand end of the graft was sutured as medially as possible on the pectoralis major muscle using number 2 Tevdek suture (Teleflex Medical) by Krackow technique. By 1-year follow-up, the patient had returned to their preinjury level of activity with symmetric strength and no complaints of cosmetic deformity.
Fascia Lata Allograft
In a case of chronic pectoralis major tear (5 years post-injury) with approximately 7 cm of tendon retraction and 1.5-cm residual gap after attempted mobilization for primary repair, a fascia lata allograft was utilized for reconstruction [32]. A Pulvertaft stitch [34] was used to suture the allograft to the tendinous portion of the pectoralis major, and three suture anchors were placed in the humerus. A suture limb from both the proximal and distal anchors were used to perform a running Krackow stitch to reduce the graft to the footprint. All additional suture limbs were passed through the allograft tissue in a simple fashion to allow for tensioning of the repair. This technique allowed for external rotation to 70° before significant tension on the repair was appreciated. At 18-month follow-up, the patient had returned to unrestricted activity with full strength and no sensation of instability.
Achilles Tendon Allograft
In a retrospective review of 11 patients who underwent pectoralis major tendon reconstruction with Achilles tendon allograft for chronic (average 12.2 months from injury) injury, three double-loaded 2.8-mm Q-Fix all-suture anchors (Smith and Nephew) were used in a triangular configuration at the anatomic footprint on the humerus to achieve reconstruction [28]. The tendinous allograft was laid over the surface of the pectoralis major muscle with a minimum of 4-cm coverage, and sutured with number 2 high-strength Orthocord sutures (Depuy Mitek) using a quilt technique with multiple lines of stitching. The suture limbs from the anchors were then sutured into the humeral end of the graft using a modified-whip-stitch technique to approximate the graft to the humeral footprint. With this technique, all patients were able to return to pre-injury levels of activity, however subjective outcomes varied as two patients reported no improvement of pain compared to preoperative levels, and three patients reported no improvement of strength after the surgery.
Zacchilli et al. [26] described three cases of chronic pectoralis major tendon reconstruction using Achilles tendon allograft. In these cases, traction sutures were utilized to allow for circumferential release and maximum mobilization of the pectoralis musculotendinous unit prior to reconstruction. A hand-held burr or narrow rongeur was utilized to create small troughs for three Corkscrew suture anchors (Arthrex, Corkscrew) that were preloaded with number 2 high-tensile suture. These suture limbs were secured to the distal end of the Achilles tendon allograft. At this time, the arm was adducted and internally rotated with tension on the aforementioned traction sutures to approximate the musculotendinous unit and allow the proximal end of the allograft to be passed over the pectoralis muscle. Using a Krackow technique, a number 5 Ethibond suture was used to suture the graft to the underlying pectoralis major muscle. Overall the technique resulted in "excellent" and "good" outcomes per the Bak criteria.
Joseph et al. [29] utilized an Achilles tendon allograft in a young patient with chronic pectoralis major tear in which there was a 3cm residual gap after circumferential mobilization of the musculotendinous unit. The allograft was sutured into the stump at the musculotendinous junction, followed by direct repair to the humeral footprint using three metal suture anchors. This patient was able to return to sporting activities 5 months after surgery and expressed no issues with functional limitations or cosmesis at 18-month follow-up.
Dermal Allograft
Dermal allograft augmentation is one of the most studied methods of surgical reconstruction of pectoralis major tears [8,16]. Neumann et al. [16] conducted a retrospective review of 19 cases of pectoralis major reconstruction utilizing dermal allograft augmentation on patients over a year from initial injury. In these cases, the dermal allograft was sutured to the native pectoralis major tendon and muscle using two number 5 FiberWire sutures in Krackow fashion. Humeral fixation at the anatomic footprint included cortical buttons (Pec Button, Arthrex; 42.1%), Biocomposite SwiveLock or Biocomposite FT suture anchor (Arthrex; 47.4%) or a combination of cortical buttons or suture anchors (10.5%). Their review showed significant improvement at follow-up for these patients on the Disabilities of the Arm, Shoulder, and Hand scale (P<0.001) and the VAS (P=0.11). Stem cells (BioDFactor; Derma Sciences) were applied to the surgical site following repair in nine patients (47.4%) to improve healing potential and decrease scar formation at the pectoralis major.
The use of acellular dermal allograft (ADA) as an interpositional graft to decrease tension on an attempted repair showed success in 2 patients with chronic pectoralis major tears [8]. In this technique, the ADA was placed so that it overlapped the tendinous stump by 2 cm, with the dermal side facing away from the tendon. The ADA was reinforced to the tendon and associated muscle belly using large nonabsorbable interrupted sutures (No. 2 Force Fiber, Wright Medical). The ADA was then anchored to the humeral footprint using three PITON suture anchors (Tornier Inc.) with suture limbs that were woven to the ADA using a running modified Becker technique [35]. At final follow-up, both patients had positive functional outcome scores, improved strength, and were able to return to work and recreational activities. Objective strength testing compared to the contralateral side showed preservation of 88% (patient 1) and 110% (patient 2).
Bone-Patellar Tendon
BPT autograft reconstruction was utilized by Zafra et al. [13] in two patients with chronic irreparable pectoralis major tendon tear, with injury occurring 10–12 months prior to operative management [13]. With the arm positioned at 50°–60° of abduction, the bone block was inserted into the humerus at the anatomic footprint and secured using a 4.5-mm cortical screw with a washer. The patellar tendon end of the graft was sutured to the musculotendinous junction. Suture technique and configuration at the graft-tendon junction were not further described in this case series. Both patients were able to resume normal recreational activities at 6 months postoperative, with good functional and subjective outcomes noted at 1 year. One patient reported unsatisfactory cosmetic results due to hypertrophic scar formation at the surgical site.
REHABILITATION PROTOCOLS
Rehabilitation protocols following chronic pectoralis major reconstruction varied slightly from study to study (Table 4) [8,13,16,26-33]. However, protocols generally followed a similar progression. Postoperatively patients are recommended to keep the affected arm immobilized in at least a sling for 3–6 weeks to allow for initial healing, but pendulum exercises are often allowed during this period. After, patients should progress to passive and active ROM exercises, followed by isometric strengthening more than 10 weeks after the procedure. This includes rotator cuff strengthening exercises, scapular stabilization and light resistance strengthening. With the goal of slow return to full activity, heavy weight training and contact sports should be postponed until at least 6 months postoperatively.
COMPLICATIONS
The rates of complications in primary pectoralis major repairs and reconstructions has been previously reported to range between 1.78% and 23%, although they are not clearly defined for chronic pectoralis major tears [6,36]. Caucasian race and chronicity (surgery more than 6 weeks from injury) were found to be the most significant risk factors for postoperative complications [37]. With nonoperative management, possible complications include persistent weakness, cosmetic deformity, hematoma, abscess formation, and myositis ossificans. Complications of operative management include infection, hypertrophic scar formation, stiffness, residual pain, decreased muscular strength, adhesive capsulitis, and re-rupture [1,38]. Further research is needed to address complications that are specific to chronic pectoralis major tears.
CONCLUSIONS
Chronic pectoralis major tears present a unique challenge to treating orthopedic surgeons due to poor tissue quality, altered anatomy, and marked tendon retraction. There are several case reports and small trials in the literature regarding management of these injuries, but no large cohort studies. While various graft types and reconstruction techniques have been found to provide successful outcomes in patients requiring reconstruction, prospective and randomized control trials are needed to determine whether there is a superior technique or graft type that should be utilized during these procedures.
Notes
Author contributions
Conceptualization: JRG, BK, RRC. Data curation: JRG, BK, BH, JG. Formal analysis: JRG. Investigation: JRG. Methodology: JRG. Project administration: JRG. Resources: JRG. Supervision: JRG, RT, RRC. Writing – original draft: JRG., BK, BH, JG. Writing – review & editing: JRG., BK, RT, RRC.
Conflict of interest
None.
Funding
None.
Data availability
None.
Acknowledgments
None.