Does physiotherapy after rotator cuff repair require supervision by a physical therapist?: a meta-analysis
Article information
Abstract
Background
A supervised physiotherapy program (SPP) is a standard regimen after surgical rotator cuff repair (RCR); however, the effect of a home-based exercise program (HEP), as an alternative, on postoperative functional recovery remains unclear. Therefore, the purpose of this meta-analysis was to compare the functional effects of SPP and HEP after RCR.
Methods
We searched electronic databases including Central, Medline, and Embase in April 2022. The primary outcomes included the Constant score, American Shoulder and Elbow Surgeons score, University of California Los Angeles shoulder score, and pain score. Secondary outcomes included range of motion, muscle strength, retear rate, and patient satisfaction rate. A meta-analysis using random-effects models was performed on the pooled results to determine the significance.
Results
The initial database search yielded 848 records, five of which met our criteria. Variables at 3 months after surgery were successfully analyzed, including the Constant score (mean difference, −8.51 points; 95% confidence interval [CI], −32.72 to 15.69; P=0.49) and pain score (mean difference, 0.02 cm; 95% CI, −2.29 to 2.33; P=0.99). There were no significant differences between the SPP and HEP. Other variables were not analyzed owing to the lack of data.
Conclusions
Our data showed no significant differences between SSP and HEP with regard to the Constant and pain scores at 3 months after RCR. These results suggest that HEP may be an alternative regimen after RCR.
Level of evidence
I.
INTRODUCTION
Rotator cuff tears lead to various clinical conditions, including muscle weakness and shoulder dysfunction. Conservative treatment is the first choice for patients with cuff tears and includes non-steroidal anti-inflammatory drug administration, steroid injection, hyaluronic acid injection, physical therapy, and exercise therapy [1]. Rotator cuff repair (RCR) is indicated for patients with rotator cuff tear when conservative treatment is unsuccessful [2]. Factors affecting RCR include tear size, symptom duration, night pain, history of trauma, and limitations in activities of daily living [3]. The primary goal of RCR is to improve shoulder functions such as range of motion (ROM) and muscle strength and to provide pain relief. RCR is traditionally followed by a supervised physiotherapy program (SPP). SSP is usually customized to the patient with RCR and includes ROM exercises, strength of rotator cuff muscles, regulation of thoracic posture, and correction of scapula position [4]. A previous study has reported that SPP after RCR allows patients to achieve a successful ROM and strength during short-term follow-up (6–12 weeks) [5]. Similarly, in medium- to long-term follow-up, acceptable improvements in function and health status have been shown in patients who underwent SPP after RCR [6,7]. These results suggest that SPP plays an important role in successful outcomes after RCR.
The effectiveness of a home-based exercise program (HEP) has been previously examined as an alternative to SPP after RCR. Roddey et al. [8] reported no significant difference in the shoulder pain and disability index at 1 year postoperatively in RCR patients who received videotape- or therapist-provided HEP instruction, suggesting that videotaped instruction may be an alternative to therapist instruction. A previous review examining the cost-effectiveness of treatment after RCR has shown that HEP using a videotape reduces the financial burden on patients [9]. Büker et al. [10] compared short-term functional results in patients with RCR who received SPP or HEP and found that HEP significantly improved the postoperative Constant score, 36-item Short Form Health Survey (SF-36) score, and pain score at 12 weeks after surgery compared to SPP. In patients with medium-sized and large rotator cuff tears who underwent RCR, significantly higher postoperative re-tear rate was observed in the HEP group than in the SSP group [11].
Additionally, Longo et al. [12] compared the effects of SPP and HEP after RCR using a meta-analysis of four randomized controlled trials. They compared the level of “postoperative pain” in patients with SPP or HEP but did not find significant difference between the two groups. Recently, another randomized controlled trial by Karppi et al. [13] compared the effects of SPP and HEP on functional outcomes at the 1-year follow-up after RCR; they did not find any significant difference in Constant score or pain level between the two procedures. These results encouraged us to conduct a new meta-analysis on postoperative shoulder function in patients with RCR receiving SPP or HEP. Therefore, this study aimed to compare the effects of SPP and HEP on functional outcomes after RCR, using a meta-analysis that included a newly published randomized controlled trial. We hypothesized that there would be no difference between SSP and HEP in terms of the effects on postoperative shoulder function and pain.
METHODS
This study was a systematic review and did not require ethics approval. The study was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [14], and its systematic review protocol was registered with the UMIN (UMIN000045454). We searched the Central, Medline, and Embase electronic databases with no limitations concerning year of publication or language; all databases were searched in April 2022. The main search terms included rotator cuff, RCR, arthroscopy, rehabilitation, physiotherapy, and exercise therapy. The search strategies used for each database can be seen in Supplementary Material 1.
The inclusion criteria for the analyzed study were as follows: studies (1) designed as randomized controlled trials, (2) that compared the functional outcome between SPP and HEP in patients who underwent RCR, and (3) that evaluated at least one outcome measure. The exclusion criteria were as follows: studies (1) that were not designed as randomized controlled trials, (2) that addressed a single type of intervention, and (3) that included patients with other shoulder disorders (e.g., rotator cuff tendinopathy, fracture, shoulder impingement syndrome, and hemiplegic shoulder).
Two authors (MK and TO) independently screened the titles and abstracts of each study identified in the search. The full texts of the studies that qualified after the first screening were further evaluated according to predefined inclusion and exclusion criteria. The two authors compared their lists, and any differences in opinion were resolved by discussion. If a consensus was not reached, a third author arbitrated the decision (JK). References from eligible articles were searched to ensure a comprehensive survey of the relevant literature.
The primary outcomes were Constant score, American Shoulder and Elbow Surgeons (ASES) score, University of California Los Angeles (UCLA) shoulder score, and pain score (visual analog scale). Secondary outcomes were ROM, muscle strength, retear rate, and patient satisfaction rate.
After selecting the final list of articles, we extracted the mean and standard deviation (SD) of the clinical outcomes. When SDs were missing, we contacted the corresponding authors for details of the clinical results. If the corresponding authors could not be contacted or did not respond, we calculated the SDs from standard errors (SEs), 95% confidence intervals (CIs), or P-values.
The risk of bias in each study was assessed using the Risk of Bias 2 (RoB2) tool by two independent authors (MK and OT) [15]. The RoB2 tool consists of five domains: randomization, deviation from intended intervention, missing outcome data, measurement of outcome, and selection of reported result. The risk of bias for each domain was evaluated as low risk, some concerns, or high risk using an algorithm with several signaling questions. Overall, a low risk of bias was recorded when the study was judged to have a low risk of bias for all domains; some concerns were recorded when the study was judged to have some concerns in at least one domain, and a high risk of bias was recorded when the study was judged to have a high risk of bias in at least one domain. Any disagreements were resolved through discussion or consultation with another author (JK).
We pooled clinical outcomes with similar characteristics (outcome measures and timing of outcome measurements) to compare the effects of SPP and HEP. All analyses were performed using Review Manager 5.4 for meta-analyses using mean differences and 95% CIs. We used random-effects models for all analyses. For a P<0.05, the effect was considered significant. Additionally, I2 statistics were used to measure statistical heterogeneity among the trials in each analysis. We interpreted the I2 statistic as follows: 0%–40% not important, 30%–60% moderate heterogeneity, 50%–90% substantial heterogeneity, and 75%–100% considerable heterogeneity.
RESULTS
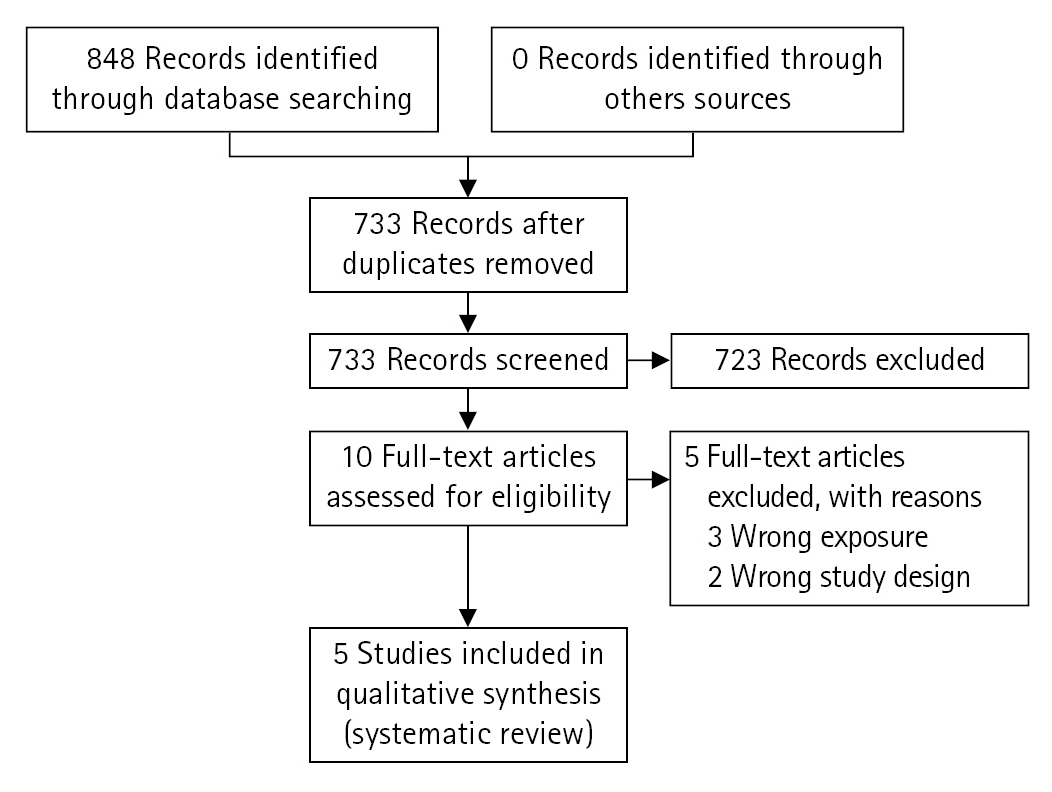
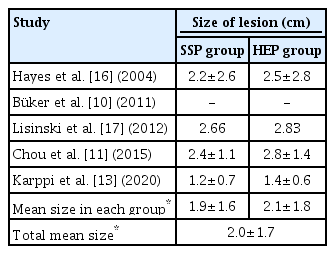
The search yielded 848 records from three databases. After removing duplicates, 733 records remained. We ultimately screened 10 full-text articles and identified five studies that were included in the review (Fig. 1). A total of 212 patients (119 men and 93 women; mean age, 57.4 years) were included in the five studies, with an average follow-up duration ranging from 5 weeks to 1 year (Table 1). The mean tear size was identified in four studies: 1.9±1.6 cm in the SSP group and 2.1±1.8 cm in the HEP group. The total mean size was 2.0±1.7 cm (Table 2).
Five studies [10,11,13,16,17] used three surgical techniques: open deltoid splitting in 58 patients (27.4%), mini-open technique in 52 patients (24.5%), and arthroscopic technique in 102 patients (48.1%). The patients were categorized into two groups: SPP (104 patients) and HEP (108 patients). The SSP group performed active or active-assisted shoulder exercises, scapular retraction, pendulum exercises, shoulder muscle strengthening with an elastic band, myofascial release, proprioceptive neuromuscular facilitation, or rotator cuff stretching. The frequency of SPP varied among studies: daily [17], once a week [16], 5 days per week [10,11], and once in 2 weeks [13]. Patients in the HEP group were given oral and written descriptions of their treatments. The HEP group performed active or active-assisted shoulder exercises, pendulum exercises, progressive resistive strengthening, and physical therapy (e.g., ice and moist heat). In Büker et al. [10], the patients received weekly exercise management by a physical therapist, although most of the interventions consisted of home exercise; this study was allocated to the HEP group. In other studies, patients received only HEP [11,13,16,17].
Details of the risk of bias judgements for the primary outcomes of the included studies are presented in Fig. 2. In the domain of randomization, one study had a low risk of bias [13], two studies had some concerns [10,11], and two studies had a low risk of bias for missing outcome data [10,11]. All studies [10,11,13,16,17] had a high risk of bias for deviations from intended interventions and overall bias.
The timing of outcome assessment varied at 20 days (one trial) [17], 40 days (one trial) [17], 6 weeks (one trial) [16], 3 months (four trials) [10,11,13,16], 6 months (one trial) [16], and 1 year (one trial) [13] after surgery. Three studies [10,11,13] described the Constant score at 3 months, of which 104 patients (52 SPP and 52 HEP patients) from two studies [10,13] were included in the meta-analysis. One study [11] was excluded from the analysis because it measured only the muscle strength items of the Constant score. Finally, analysis of two of the five studies showed that the Constant score of the SPP group was not significantly different from that of the HEP group (mean difference, − 8.51 points; 95% CI, −32.72 to 15.69; P=0.49; I2= 95%) (Fig. 3).
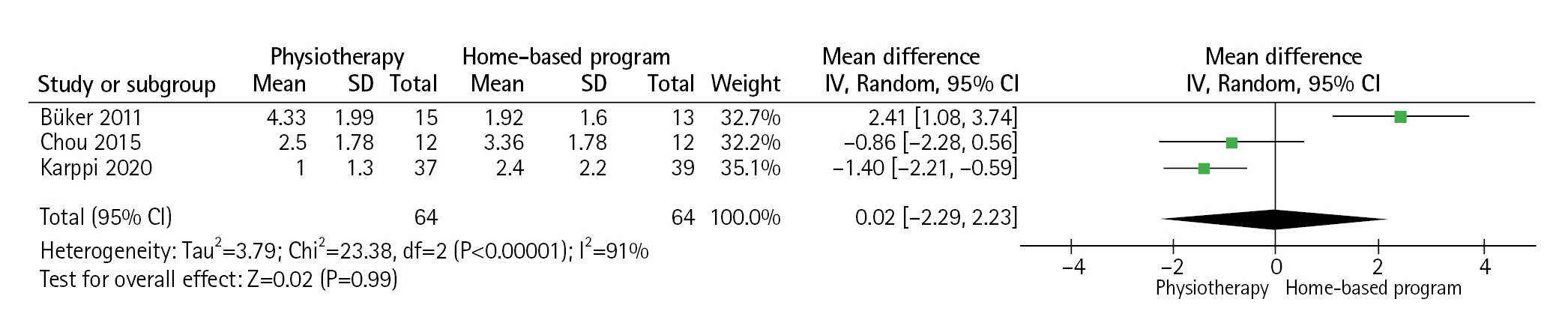
Four studies [10,11,13,17] described the pain score. Of these, 128 patients (64 SPP and 64 HEP patients) from three studies [10,11,13] who had available pain scores at 3 months postoperatively were included in the meta-analysis; one study [17] in which the assessment timing (20 and 40 days postoperatively) did not match that of the other studies was excluded. The analysis showed that the pain score of the SPP group at 3 months after surgery was not significantly different from that of the HEP group (mean difference, 0.02 cm; 95% CI, −2.29 to 2.33; P=0.99; I2=91%) (Fig. 4).
The other variables (e.g., ASES and UCLA scores) were not analyzed owing to lack of data. These included one study [11] investigating the ASES and UCLA scores, two studies [16,17] evaluating ROM with different measurement points and items, one study [16] evaluating muscle strength, and one study [11] evaluating the retear rate.
DISCUSSION
RCR is an operative procedure that improves function and relieves pain when conservative treatments fail. Optimal postoperative rehabilitation is necessary to reduce postoperative stiffness and pain. Patients usually undergo SPP after RCR; however, there is growing evidence that HEP is a useful regimen after surgery. In 2020, a meta-analysis compared SPP and HEP after RCR and demonstrated no significant difference in postoperative pain score between the two procedures [12]. By updating a new randomized clinical trial comparing the functional outcome between SPP and HEP, the present meta-analysis successfully demonstrated no significant difference not only in postoperative pain score, but also in the Constant score at 3 months after surgery. Thus, our data showed HEP as an acceptable alternative procedure to SPP after RCR.
Charousset et al. [18] have shown that functional recovery is accelerated from 3 months after RCR, progresses until 12 months, and then reaches a plateau. Iannotti et al. [19] identified the 3-month time point after surgery as one of the critical time points for recovery in patients with RCR. In the present study, the Constant score, frequently used as an evaluation tool for functional outcomes after RCR [20], showed no statistically significant difference between SPP and HEP at 3 months after surgery. Although we did not compare the Constant score beyond 3 months after surgery because of lack of data, these results indicate that the effect of HEP on functional recovery after RCR is comparable to SPP in the short-term period after surgery.
A previous meta-analysis showed no significant difference in pain scores after RCR between SSP and HEP, although the timing of outcome assessments was not uniform in (40 days to 3 months) [12]. The present study adjusted the assessment timing to 3 months after surgery and evaluated the pain score during this period. Consequently, there was no significant difference in pain scores between the two procedures, consistent with previous results. Karppi et al. [13] reported that the pain score was not significantly different between the SPP and HEP groups 1 year after RCR. Taken together, these findings indicate that HEP is a useful regimen after surgery and is comparable to SPP.
Büker et al. [10] compared the cost and clinical results of SPP with HEP in light of a cost-effective postoperative regimen for RCR. Their study reported significantly lower Constant score and SF-36 and significantly higher pain score after RCR in the SPP group, in addition to a total cost approximately twice that of the HEP group. In 2017, Dickinson et al. [9] conducted a systematic review of the cost-effectiveness of SPP and HEP but did not demonstrate significant differences between them because of limited data. Cost-effectiveness was not analyzed in our study and should be clarified in future studies.
The limitations of the present study are as follows. First, it did not analyze all important clinical variables (e.g., functional scores in addition to Constant score, surgical techniques, tear sizes, coexistence of contracture, and retear rate). Second, group analysis subdivided by tear size/type was not performed because of lack of data. Third, the present study did not show clinical outcomes for a period longer than 3 months postoperatively. A strength of our study is that we offered new insights into the comparison between SPP and HEP after RCR by exclusively analyzing randomized controlled trials with a high evidence level.
CONCLUSIONS
Using a meta-analysis, we compared functional outcomes between SSP and HEP groups. Our data showed no significant difference in Constant or pain score at 3 months after RCR between the two groups. Therefore, we conclude that HEP may be an alternative regimen to the typical SSP after RCR.
Notes
Author contributions
Conceptualization: TO. Formal analysis: KN, MS. Methodology: TO. Project administration: MK, MG. Supervision: JK. Writing – original draft: MK. Writing – review & editing: MK, MG, HN, HO.
Conflict of interest
None.
Funding
None.
Data availability
Contact the corresponding author for data availability.
Acknowledgments
None.
SUPPLEMENTARY MATERIALS
Supplementary materials can be found via https://doi.org/10.5397/cise.2022.01410.
Final list of unique webpages included in our study