Implant selection for successful reverse total shoulder arthroplasty
Article information
Abstract
Reverse total shoulder arthroplasty (RTSA) emerged as a new concept of arthroplasty that does not restore normal anatomy but does restore function. It enables the function of the torn rotator cuff to be performed by the deltoid and shows encouraging clinical outcomes. Since its introduction, various modifications have been designed to improve the outcome of the RTSA. From the original cemented baseplate with peg or keel, a cementless baseplate was designed that could be fixed with central and peripheral screws. In addition, a modular-type glenoid component enabled easier revision options. For the humeral component, the initial design was an inlay type of long stem with cemented fixation. However, loss of bone stock from the cemented stem hindered revision surgery. Therefore, a cementless design was introduced with a firm metaphyseal fixation. Furthermore, to prevent complications such as scapular notching, the concept of lateralization emerged. Lateralization helped to maintain normal shoulder contour and better rotator cuff function for improved external/internal rotation power, but excessive lateralization yielded problems such as subacromial notching. Therefore, for patients with pseudoparalysis or with risk of subacromial notching, a medial eccentric tray option can be used for distalization and reduced lateralization of the center of rotation. In summary, it is important that surgeons understand the characteristics of each implant in the various options for RTSA. Furthermore, through preoperative evaluation of patients, surgeons can choose the implant option that will lead to the best outcomes after RTSA.
Level of evidence: IV.
INTRODUCTION
Reverse total shoulder arthroplasty (RTSA) was a paradigm-shifting concept in shoulder arthroplasty. While conventional arthroplasty focused on restoring the anatomical structure as much as possible, RTSA was designed to replace the biomechanical function of torn rotator cuffs [1-3]. The current concept of RTSA was first introduced by Grammont in 1985 on the basis that the deltoid could compensate for the function of a torn rotator cuff and stabilize the shoulder joint. [4] Therefore, the center of rotation (COR) was moved in a medial and distal direction. The medialized COR allowed the deltoid muscle to be used for increased forward flexion and abduction, and the distalized COR lengthened the moment arm of the deltoid for better function (Fig. 1) [5-8].
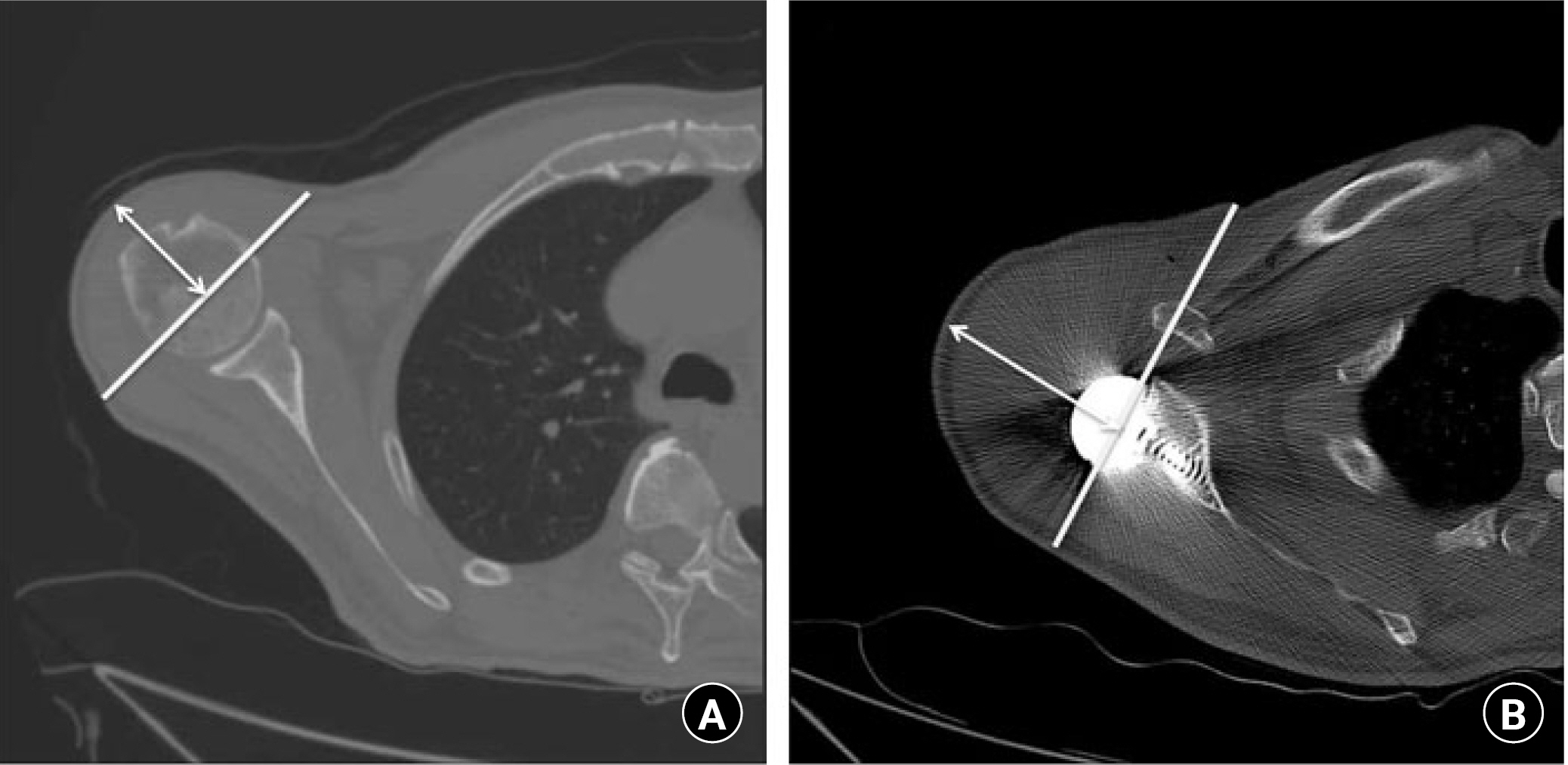
Effect of a medialized center of rotation. With a medialized center of rotation, the proportion of the deltoid muscle from the center of rotation is much larger, as seen in the postoperative image. Axial images of (A) preoperative and (B) postoperative computed tomography scan.
Early implants for RTSA were composed of a keel or peg-type baseplate and inlay designed humeral component. The baseplate initially was fixed with cement for high stability, and cement was also used for fixation of the humeral component. However, cemented fixation had drawbacks such as loss of bone stock and difficulty in revision. To avoid possible future bone loss, cementless implants were introduced. The glenoid baseplate was fixed more securely with a central screw and several peripheral screws, and the humeral component was press-fit to the metaphysis. For greater bone preservation, a shorter stem length and a stemless design of the humeral component emerged. Furthermore, the concept of lateralization was presented to overcome problems from medialization of the COR such as loss of shoulder contour, reduced stability of the artificial joint, weaker external and internal rotation power, and scapular notching [9-12]. However, the lateralized implant could decompensate the advantages of medialization of the COR. In addition, excessive lateralization induced complications such as stress fracture of the scapular spine and/or subacromial notching provoked by impingement at abduction [13]. Therefore, the advantages of medialization and lateralization should be balanced to improve the outcome. In this review article, we will discuss the design concept of current implant options for RTSA and suggest optimal implant selection for successful outcome in patients.
IMPLANT DESIGN AND SURGICAL TECHNIQUE
Glenoid Component
After introduction of the Grammont-type implant, the design of the RTSA has been improved through modification. A central post and screw fixation have enabled more stable fixation of the baseplate with optional peripheral screw fixation. Also, variations in baseplate size, material, and peripheral screw configuration were developed to achieve customization to patient anatomy.
Inappropriate positioning of the glenoid baseplate is one of the most common causes of complications with poor long-term outcomes. For better positioning of the baseplate, precise preoperative planning is important. The size and version of the glenoid and the amount of glenoid bone stock can be measured with computed tomography (CT). The size of the glenoid is known to be correlated with the height of the patient [14,15], and previous studies have found that the size of the glenoid in the Asian population is smaller than that of the North American population [14]. Therefore, several studies have argued the necessity of a smaller baseplate for Asian populations [16,17]. For stable baseplate fixation, central fixation plays a key role and can be achieved with a monoblock baseplate screw, central peg, central post, or modular central screw [18]. The modular central screw is widely used for the convertible glenoid option and provides similar stability to the central post [3].
The center of the baseplate can be fixed with a central screw or peg, and the baseplate in the modern design offers the option of two to six peripheral screws to increase stability. The number and/or length of peripheral screws affect the stability [19]. Usually, the superior screw should be headed to the base of the coracoid process anteriorly, and the inferior screw should locate to the scapular neck postero-inferiorly for better strength of fixation [19,20]. Improper insertion of peripheral screws could provoke suprascapular nerve injury and/or scapular fracture. Suprascapular nerve injury can occur if superior or posterior screws are too long and head toward the suprascapular notch. Also, if superior and posterior screws penetrate the scapular spine, they can act as stress risers and lead to scapular fracture. Therefore, the length and direction of superior and posterior screws should be checked carefully [21-23]. An oval-shaped baseplate could be better for fixation, but it can be too large for Asian patients with a relatively smaller-sized glenoid [14,20,24]. Circular baseplates are technically better for altering superior and inferior offsets [25].
If a glenoid bone defect is noted preoperatively, eccentric reaming, augmented baseplate, or bone graft could be considered based on defect severity [26,27]. Eccentric reaming can correct mild glenoid erosion, a glenoid version within 15°. However, in the case of severe glenoid erosion, eccentric reaming can remove quite a large amount of glenoid bone stock and eventually decrease the stability of implants. Therefore, for severe glenoid bone defect, eccentric reaming is not an appropriate option, and augmented baseplate and/or bone graft should be considered. An augmented baseplate can be used for a large bone defect, but sufficient bone stock is still needed for fixation of the baseplate. Furthermore, a specialized reamer with an appropriate guide pin position is needed for preparation of the glenoid when using an augmented baseplate [18]. Although some studies have shown favorable outcomes with augmented baseplates, there is a lack of long-term outcome data [28,29]. For more significant bone loss, a bone graft might be the only option as it can support the baseplate even in a large bone defect. However, bone graft involves technical difficulty and risk of graft resorption and subsidence [30].
Scapular notching was one of the critical complications in the conventional RTSA and could lead to glenoid baseplate loosening. Glenoid position and individual anatomical characteristics were thought to be risk factors of scapular notching. Scapular neck length (SNL) is a risk factor of scapular notching, with a length less than 9 mm showed a greater risk [31]; therefore, surgeons should consider lateralization of the glenoid component in such patients. A more detailed explanation of the lateralized glenoid (LG) component is described below. As well as a LG component, eccentric positioning with an inferior overhang of the glenoid component can be used to prevent scapular notching. Eccentric positioning of the glenoid component can be achieved by inferior translation and/or inferior tilting of the glenoid baseplate. The inferior rim of the baseplate should be positioned along with the inferior rim of the bony glenoid. There has been some debate regarding the inferior tilt of the glenoid baseplate. However, considering the normal anatomy of the glenoid with a 5° superior tilt and the superior wear of cuff tear arthropathy, a 10° inferior tilt of the baseplate is recommended to prevent scapular notching [32,33]. A glenosphere with an inferior offset (inferiorly overhanging glenosphere) can also be used for inferior translation of the glenoid component [34-37]. Several studies have shown that inferior overhang of the glenosphere provided a better range of motion (ROM) with a significantly lower rate of scapular notching [38-41]. For the Korean population, an inferior overhang of about 2.9 mm resulted in a lower rate of scapular notching with superior clinical outcomes [21].
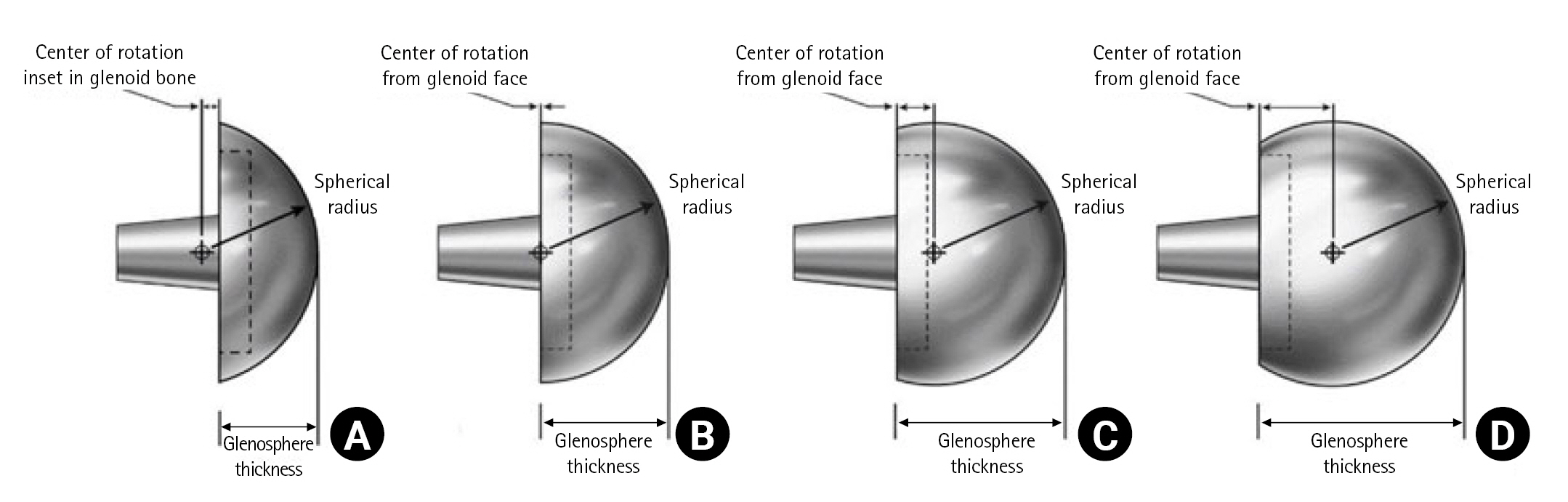
As we described above, the concept of the LG component emerged as a solution to prevent scapular notching. However, a LG implant does not mean that the COR is lateralized compared to the anatomical COR. It means that the COR is lateralized compared to conventional medialized implants and remains medial to the original COR (Fig. 2) [42,43]. Lateralization can be obtained by adjusting implant options or surgical techniques. Implants with these characteristics are known as LG implants, and conventional glenoid implants are medialized glenoid (MG) implants. The LG implant also has the advantage of recovering the original shoulder contour by moving the COR laterally. With the LG implant, further stability can be achieved by increasing the deltoid wrapping angle. However, the LG could decompensate the function of the deltoid and weaken forward flexion. For patients with pseudoparalysis, this could be disadvantageous [44,45].
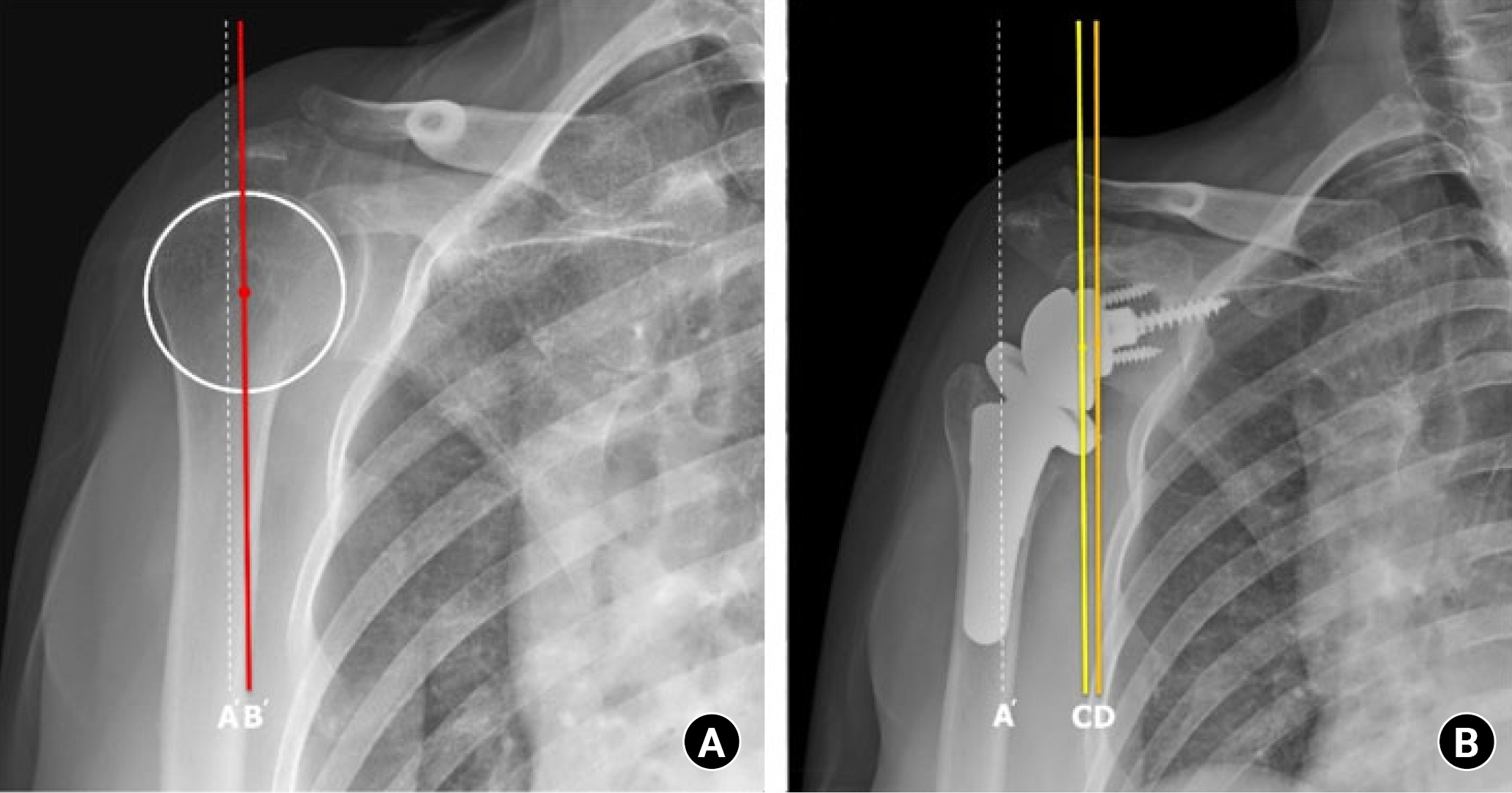
Center of rotation of the lateralized implant remains medial to the original center of rotation (COR). (A) Preoperative X-ray. (B) Postoperative X-ray. A’: lateral margin of the acromion, B’: anatomical COR, C: COR in lateral glenoid implant, D: COR in medial glenoid implant.
A thicker glenosphere can lateralize COR relatively easily. However, a thicker lateralized glenosphere theoretically increases the shear force on the glenoid baseplate interface and can cause excessive movement and loosening of the glenoid component. However, several computer simulations and cadaveric biomechanical studies have reported that a 1 cm thicker glenosphere was not related to meaningful loosening of the baseplate because the shoulder is not a weight-bearing joint [46]. Therefore, before using a thicker lateralized glenosphere, this complication should be considered (Fig. 3) [47,48]. A glenosphere with a larger diameter also can maintain the tension of rotator cuffs and improve deltoid muscle coverage. However, insertion of a larger glenosphere is technically demanding in patients with tight joint space or stiff shoulders and its use is limited in the relatively small sized joints in Asian populations. Also, a glenosphere with a larger diameter is reported to increase the volumetric wear rate of the polyethylene liner [49].
A bone graft under the baseplate can also be used for lateralization. A thick and hard strut bone graft can be inserted between the reamed native bone and baseplate and can be firmly fixed to the baseplate using central and peripheral screws. Although a bone graft is technically more difficult, it can provide enough bone stock with less shear force than the thicker glenosphere even in patients with a large bone defect. It is important to achieve graft union with stable fixation because malunion or absorption of bone graft can occur postoperatively [50]. Both autograft and allograft can be used with comparable outcomes, and the osteotomized humeral head can be used as an autograft for the bone graft [51].
Humeral Component
A conventional Grammont-type humeral implant is composed of a long humeral stem with an inlay design, and the humeral tray is seated inside the metaphysis. Inlay humeral stems are fixed with cement from the metaphysis to the diaphysis. A cemented fixation could provide stability immediately after surgery. However, to insert the same size of humeral stem, a larger amount of metaphyseal bone stock should be reamed for cemented fixation compared to cementless press-fit fixation. Furthermore, cement could cause complications such as radial nerve palsy, which is induced by thermal damage to peripheral soft tissue from cement leakage [52]. Moreover, in a revision scenario, extraction of the cemented stem could increase the risk of fracture or the amount of bone loss during removal of the cement mantle. As a result, cementless fixation of the humeral component has been developed, and recent research showed satisfactory long-term outcomes [53].
To preserve humeral bone stock, a shorter humeral stem has been introduced. Humeral stems with lengths shorter than the conventional long stem also could reduce stress shielding, which could be a risk factor for loosening of the stem and metaphyseal bone loss [54]. Several studies have reported favorable outcomes for short-stem implants compared to the conventional long-stem design [55]. However, malalignment of short stems could be more frequent than in conventional long stems [56-58], so further long-term comparative studies are needed to assess the appropriate length of humeral stems in terms of stress shielding and alignment. As shorter designs have been developed, extreme designs with stemless humeral components without diaphyseal fixation were introduced (Fig. 4). A stemless humeral component could preserve more humeral bone stock, allow easier revision surgery, and reduce stress shielding. However, similar to the short stem component, the risk of malalignment is relatively higher than in the conventional stem [59]. Although recent studies have presented encouraging outcomes with stemless design, it is a concern that studies have only reported short- to mid-term outcomes [60-63]. Furthermore, complications such as tray failure and periprosthetic fracture of the lateral cortex due to impaction of the implant have been reported (Fig. 5) [64,65].

Various types of humeral components on postoperative X-ray. All implants were from Zimmer Biomet. (A) Standard stem. (B) Mini stem. (C) Micro stem. (D) Nano stem (Stemless).
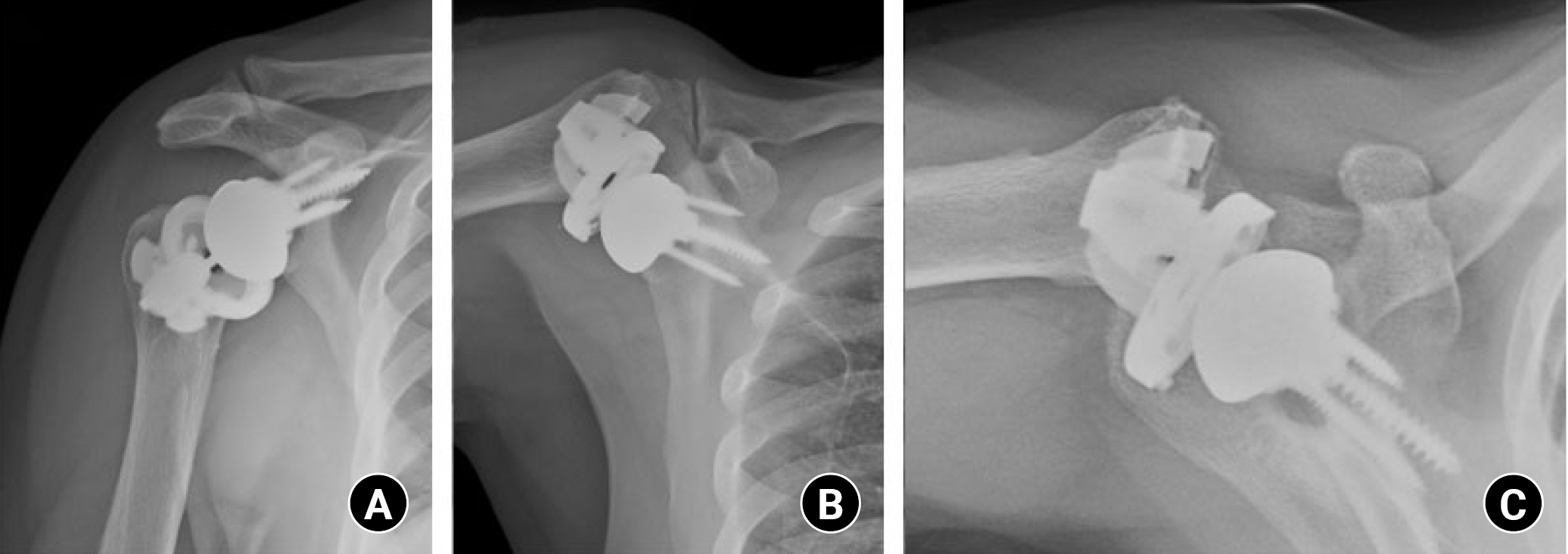
Tray failure case. A 72-year-old male patient underwent reverse total shoulder arthroplasty with a stemless humeral component under diagnosis of rotator cuff tear arthropathy. Twenty-two months after surgery, while raising his hands to scratch his nose, he felt a sudden shift with acute shoulder pain. Follow-up X-ray showed tray failure of the humeral component. (A) Shoulder 30° caudal tilt view. (B) Shoulder lateral view. (C) Shoulder axial view.
Unlike conventional arthroplasty, which focused on restoration of the original anatomy, RTSA focused on restoration of function with a non-anatomical design. Therefore, humeral retroversion was targeted to produce better functional outcomes rather than restoring normal anatomy. To improve the functional outcome, the importance of impingement-free ROM and external/internal rotation power was emphasized. Conventionally, an increased retroversion of the humeral component was known to result in the increase of external rotation [66]. Several studies have evaluated the effect of humeral retroversion on impingement-free ROM and rotational power, although there are still debates on the optimal retroversion of humeral implants [67-71]. A previous study found that 20° of retroversion yielded a better outcome than retroversion less than 20° [70]. On the contrary, other studies have argued that a 0° retroversion showed the least impingement with better ROM [69-71]. Meanwhile, cadaveric research concluded that a 20° to 40° retroversion showed a more balanced ROM of internal and external rotation [68]. However, a recent comparative clinical study showed the importance of restoring anatomic retroversion of the humerus [67]. In that study, functional outcome after RTSA was compared between a group of patients with humeral retroversion fixed at 20° and a group of individualized retroversion determined according to the native humeral version of the patient. Even though RTSA does not restore the original anatomy of the shoulder, individualized retroversion was thought to show a better outcome considering the original soft tissue balances around the shoulder. The individualized retroversion group showed better ROM in forward flexion, external rotation, and internal rotation. In addition, the pain visual analog scale (VAS) score of the individualized retroversion group was significantly lower than that of the fixed group. Considering the relatively large retroversion angle of Asian populations, individualizing a retroversion can provide better outcomes, especially with regard to the ROM (Fig. 6) [67,71,72].
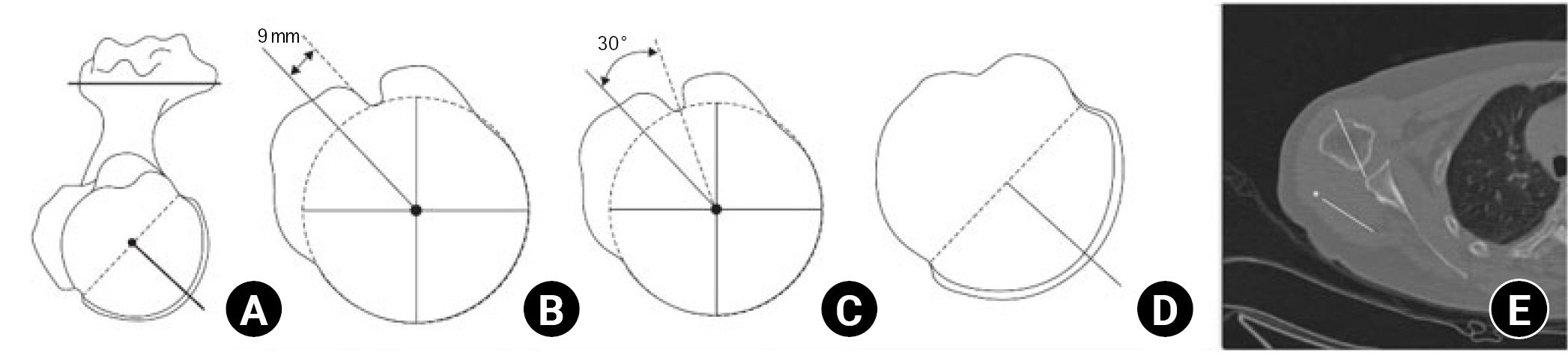
The five methods of measuring humeral retroversion. (A) Method 1 measures the angle between the perpendicular axis of the articular surface and the transepicondylar axis (TEA). (B) Method 2 measures the angle between the 9 mm posterior margin of the bicipital groove axis and the TEA. (C) Method 3 measures the angle between the bicipital groove center axis and the elbow transepicondylar axis –30°. (D) Method 4 measures the angle between the axis through the distal humeral head central axis and TEA +2.4° at the distal humeral head. (E) Method 5 measures the angle between the metaphyseal axis and the TEA +2.5°. Adapted from Oh et al. Clin Orthop Surg 2017;9:223-31 [71].
Lateralization of the implant could also be achieved on the humeral side. A lateralized humeral (LH) implant has the advantage of maintaining medialized COR, which maximizes the function of the deltoid for forward flexion as intended in the initial design of the Grammont-type glenoid implant. It can prevent loss of shoulder contour and increase the stability of the implant by improving the deltoid wrapping angle. Furthermore, tension in the remnant rotator cuff and deltoid muscle coverage can be improved compared to those of conventional medialized humeral (MH) implants [73-75]. Maintained tension in the rotator cuffs preserves internal and external rotation. Moreover, the LH implant could decrease the impingement between the humeral implant and the scapular neck and could prevent scapular notching. There are various methods of lateralizing the humeral implant. The first method is to decrease the neck-shaft angle from 155° to 135°–145°, which is closer to the anatomical neck-shaft angle. Using implants with a lower neck-shaft angle of 135°–145°, the distance between the humerus and scapular neck could be increased with the height of the humeral tray. As a result, the stem is placed in a more lateral position compared to the original design (Fig. 7). This can help to overcome the drawbacks of medialized implants and prevent scapular notching by reducing impingement [76]. Second, the onlay humeral implant instead of the conventional inlay implant can lateralize the humerus. In the conventional inlay implant, the humeral tray could be seated stably in the metaphysis. However, a large amount of metaphyseal bone loss during insertion of the implant and impingement between the humerus and scapular neck could not be avoided. On the contrary, in an onlay implant, the tray is positioned on the cutting surface of the humeral head, and it allows more bone stock to be preserved than the inlay design. Third, increasing the thickness of the polyethylene insert or the tray can also lateralize the humeral implant. However, the thick polyethylene moves the humerus not only laterally, but also distally. Also, as with the large glenosphere, it has limitations in patients with small joint space or tight shoulders.
An LH implant is not the optimal choice. Excessive lateralization of the humeral implant can induce impingement between the humerus and acromion, which can lead to subacromial notching. (Fig. 8) [77,78]. To prevent such complications happening from excessive lateralization, humerus options can be modified with a medial eccentric tray, which has a medial offset compared to the conventional concentric tray (Fig. 9). With the use of the eccentric tray, the humeral component moves more distal and medial. Unlike modifying the thickness of the polyethylene liner that moves the humeral component more distal and lateral as it thickens, the medial eccentric tray would prevent abduction impingement with medialization and overcome pseudoparalysis with distalization. Taken together, these findings suggest that a medial eccentric tray might be helpful in patients with pseudoparalysis or patients at risk for subacromial notching.
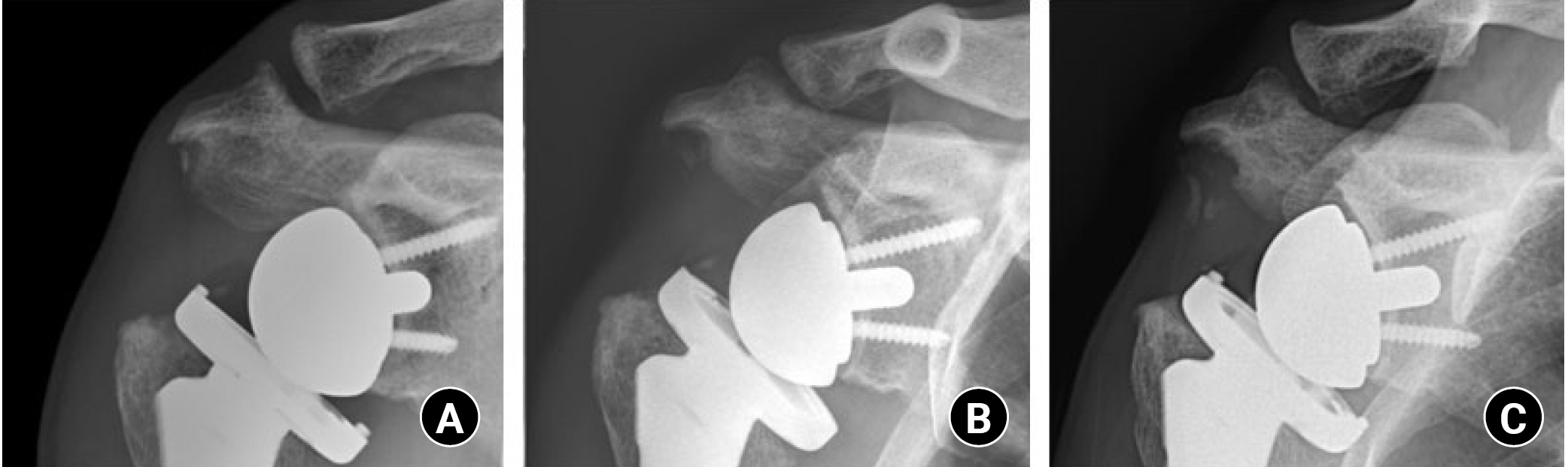
A case of subacromial notching with a lateralized humeral implant. In postoperative 3-month (A), 6-month (B), and 1-year (C) plain radiograph images, progressive subacromial bone resorption is noted.
MEDIAL AND LATERAL IMPLANT OPTIONS IN RTSA
A conventional Grammont-type design was successful in compensating for the function of torn rotator cuffs [73,76,79]. However, the medial and distal shifts of the COR produced several drawbacks [80]. Lateralization was expected to overcome the disadvantages of the conventional implant. However, where to lateralize, the glenoid or the humerus, and the degree of how much to lateralize remain controversial. Considering lateralization options of both the glenoid and the humerus, four combinations are possible, MG-MH, MG-LH, LG-MH, and LG-LH. The type differs from implant to implant, and surgeons must be aware of the characteristic of chosen implants (Fig. 10).

Four combinations of medial and lateral options in the reverse total shoulder arthroplasty implant. Lateralization can be performed in both glenoid and humeral implants. In these images, a thick glenosphere represents a lateralized glenoid implant, and the onlay type humeral implant represents a lateralized humeral implant. (A) Medial glenoid-medial humerus implant. (B) Medial glenoid-lateral humerus implant. (C) Lateral glenoid-medial humerus implant. (D) Lateral glenoid-lateral humerus implant.
MG-MH type implants are the classic, conventional type of RTSA. They have been used for years, and many clinical data with long-term results have been reported [81,82]. With a large moment arm of the deltoid for abduction, this type of implant is effective in patients with pseudoparalysis and increases abduction and forward elevation. However, scapular notching is more common than in other types of implants, and internal and external rotation is limited due to the decreased tension of the rotator cuff [83]. LG-MH type implants improve internal and external rotation by increasing the tension of the remaining rotator cuff. Also, scapular notching is expected to be less frequent than in MG-MH type implants. However, increased shear force at the glenoid baseplate is a concern [18].
MG-LH type implants, like LG-MH types, can improve external and internal rotation and reduce scapular notching but do not increase shear force in the glenoid baseplate [84,85]. However, subacromial notching of the acromion can occur with excessive lateralization of the humeral implant [42]. The LG-LH type has the advantage of lateralization in both the glenoid and humerus. However, it is difficult to insert this type of implant in patients with tight space or stiff joint. In addition, overstuffing can trigger wear of the polyethylene insert. Also, complications from excessive lateralization could occur such as scapular stress fracture and subacromial notching [13].
After considering the factors mentioned above, surgeons can combine options to maximize the advantages of RTSA. The author suggest that surgeons decide preoperatively whether to use medialization or lateralization. If lateralization is needed, surgeons must choose to lateralize the glenoid or humerus. Even with lateralized implants, medial eccentric tray options of the humeral component could be considered when excessive lateralization increases the risk of complications such as subacromial notching.
INDIVIDUALIZED IMPLANT SELECTION FOR RTSA–PROPOSED PRINCIPLE
The author would like to explain the decision-making process of implant selection of RTSA for a specific patient. A 70-year-old female patient visited our clinic with the chief complaint of right shoulder pain, with a VAS score of 6 points. Her shoulder pain had begun 10 years before the visit, with a history of 6 shoulder dislocations. She was a housewife, and her activity and sport levels were low. She had received one steroid injection in another clinic 1 year before the visit. Physical examination of the right shoulder revealed no gross deformity. Neer and Hawkins tests were positive, as was the Jobe test. Physical examinations assessing the subscapularis (bear hug test and belly press test) were positive, but the external rotation lag sign was negative. She did not show pseudoparalysis, with an active ROM of 150º forward flexion (160º passively), 50º external rotation (60º passively), and internal rotation at the T7 level (T7 passively).
Plain radiography revealed upward migration of the humeral head, glenohumeral osteoarthritis, and acetabulization of the acromion (Fig. 11). From an X-ray image, critical shoulder angle (CSA), rotational center to acromion distance (CAD), and SNL were measured (Fig. 11A, B, and E). Bilateral shoulder anterior-posterior (AP) stress X-ray showed fixed elevation of the humeral head, which suggests an unfavorable outcome of rotator cuff healing [86], and a tight operation field was expected (Fig. 11C and D). In ongoing research by the authors, CAD, which is the distance between a line from the deltoid tuberosity to the lateral margin of the acromion and a parallel line that cross the center of the humeral head, showed greater risk of subacromial notching at a length greater than 1.4 cm. Preoperative measurement showed 34.1º CSA, 1.0 cm CAD, and 13 mm SNL.
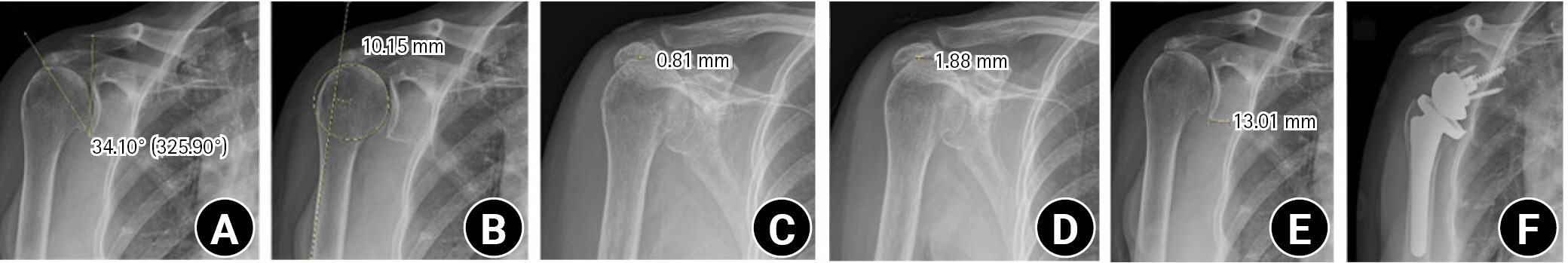
Preoperative measurements from plain radiographs and postoperative X-rays. (A) Critical shoulder angle. The value in parentheses is an angle formed between the glenoid fossa plane and a line drawn from the inferior edge of the glenoid to the lateral edge of the acromion on a true anteroposterior (Grashey) shoulder radiograph. (B) Rotational center to acromion distance. (C) Measurement of acromiohumeral distance. (D) Measurement of acromiohumeral distance with downward stress on the wrist. (E) Scapular neck length. (F) Postoperative plain radiograph.
A magnetic resonance image showed a complete tear of the supraspinatus and infraspinatus tendon with a 6.1 cm retraction and a 5.5 cm anterior-to-posterior dimension. A full-thickness tear of the subscapularis tendon and a complete tear of the biceps long head tendon were also found. Fatty degeneration was assessed as 4, 2, 0, 3 (supraspinatus, infraspinatus, teres minor, and subscapularis) by Goutallier classification (Fig. 12). The rotator cuff healing index (ROHI) was calculated as 13 points, at which the failure rate is 86.2% [48]. From preoperative CT, humeral retroversion was 28.7º (31.2º to the left) and glenoid anteversion was 2.7º (0.7º anteversion to the left) (Fig. 13).
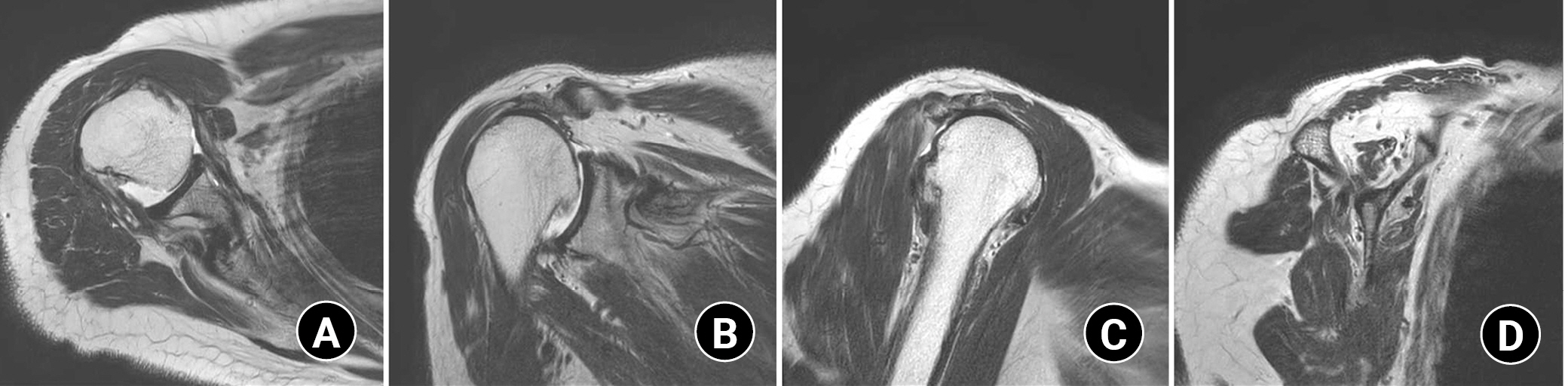
Preoperative magnetic resonance imaging image of the patient undergoing reverse total shoulder arthroplasty. It shows full-thickness tears of the subscapularis and complete tear of supraspinatus, and infraspinatus tendon. Goutallier classification of fatty degeneration was 4:2:0:3 (supraspinatus : infraspinatus:teres minor : subscapularis). (A) Axial image. (B) Coronal image. (C) Sagittal image. (D) Sagittal image for the evaluation of fatty degeneration.
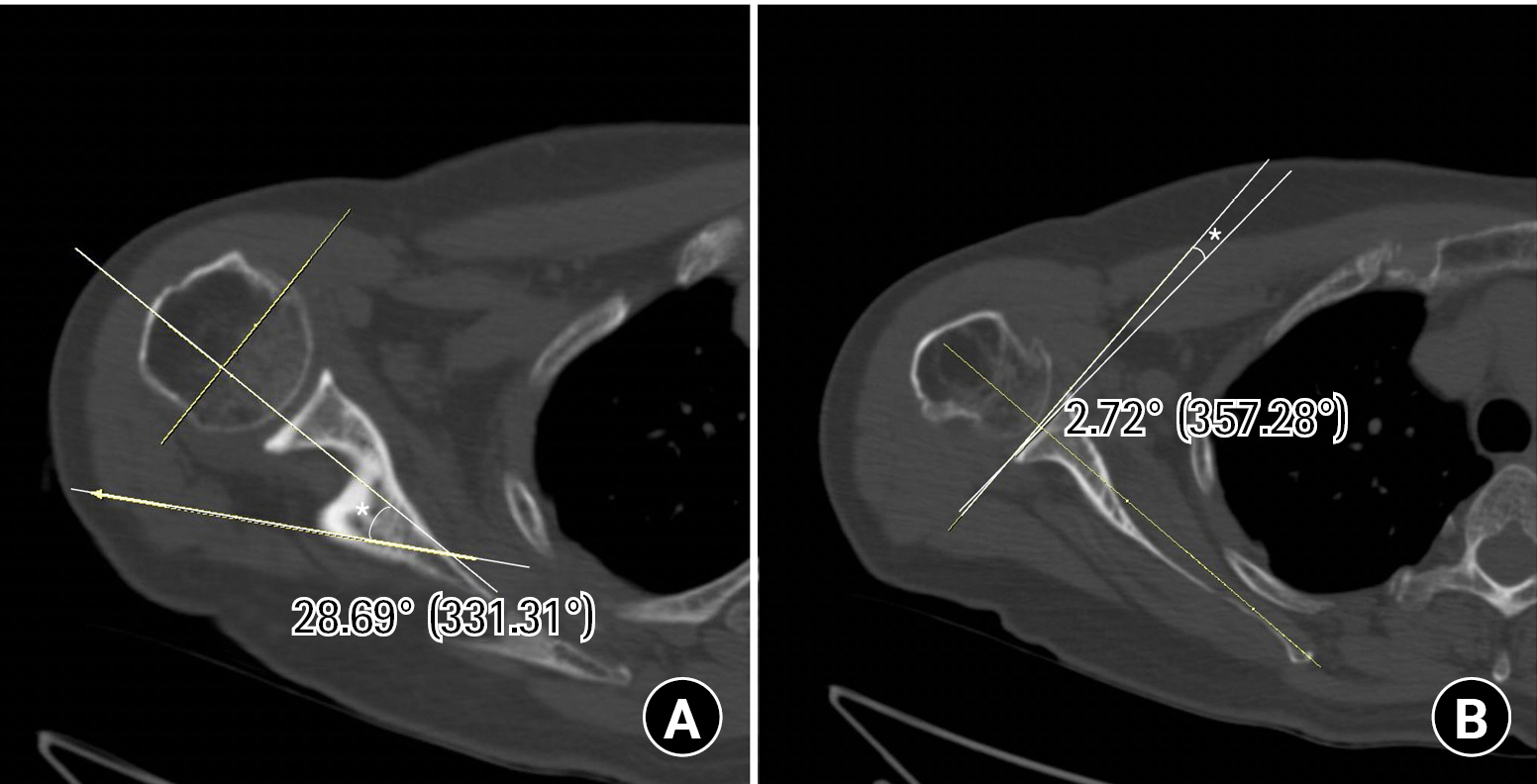
Preoperative measurement of humerus and glenoid version. (A) Humeral retroversion (asterisk, 28.69°) was measured in correlation with the transepicondylar axis of the distal humerus (elbow). (B) Glenoid version (asterisk, 2.72°) was measured using the Friedman method.
With this information, the authors followed our flow chart for implant selection using several preoperative measurements as shown as follows. (1) The patient had a massive rotator cuff tear, including the subscapularis with cuff tear arthropathy. Her ROHI was 13, and the stress X-ray showed fixed humeral head elevation. (2) A glenoid bone defect was not prominent on X-ray and CT scan. The SNL was 13 mm. (3) Humeral retroversion was measured as 28.7º (31.2º on the contralateral side) on CT scan. There was no deformity of the humeral shaft. (4) Lateralization of the implant was chosen based on preoperative measurements of 34.1º CSA and 1.0 cm CAD. The patient did not show pseudoparalysis on preoperative physical examination.
The corresponding strategies for appropriate implant selection in RTSA are as follows (Fig. 14). (1) These factors suggest a poor outcome in rotator cuff healing; therefore, RTSA was selected for this patient. Due to the fixed humeral head elevation on the stress X-ray and possible tightness of the joint, the humeral neck should be cut at a lower level than usual. The tear in the subscapularis indicated a lateral implant for stability. (2) No severe deformity in the glenoid was found, so patient-specific instrumentation was not needed, and an augmented baseplate or bone graft was not considered. The SNL was long, so a medial glenoid implant was selected and a thicker glenosphere was not necessary. To prevent scapular notching, a conventional 36-mm inferior overhang glenosphere was chosen with inferior placement of the baseplate with 10º inferior tilt. (3) A humeral implant was planned to be fixed as close as possible to the patient’s native humeral retroversion of 28.7º. Therefore, a retroversion guide of the humeral stem was set at 30º retroversion. To prevent malalignment of the humeral stem, a medium-sized (83 mm) stem was selected over a short stem. (4) Considering the absence of pseudoparalysis with a CSA larger than 32º, a lateral humeral implant would be better. A concentric tray was chosen because CAD was less than 1.4 cm. In this case, the Biomet Comprehensive implant (MG-LH) was used due to the features mentioned above (Fig. 11F).

Guide for individual implant selection in reverse total shoulder arthroplasty. CSA: critical shoulder angle, ERLS: external rotation lag sign, CAD: rotational center to acromion distance, SNL: scapular neck length, MG-MH: medial glenoid-medial humerus implant, MG-LH: medial glenoid-lateral humerus implant.
CONCLUSION
To achieve the best outcome from RTSA, understanding of the biomechanical characteristics of components is essential. A glenoid implant should be placed inferiorly with an inferior tilt to prevent scapular notching. Size and lateral offset of the glenosphere should be selected based on individual anatomical characteristics. For the humerus, short stems and stemless designs are gaining popularity, but the risk of malalignment should be considered. LH implants can be selected for patients without pseudoparalysis for a better ROM. If patients have a risk of subacromial notching, a medial eccentric humeral tray should be used. Considering these individual factors, the best implant can be chosen and will lead to the best outcomes.
Notes
Author contributions
Conceptualization: JHO. Resources: JHO. Supervision: HJJ. Writing – original draft: JHO. Writing – review & editing: HJJ, YSW.
Conflict of interest
None.
Funding
None.
Data availability
None.
Acknowledgments
None.